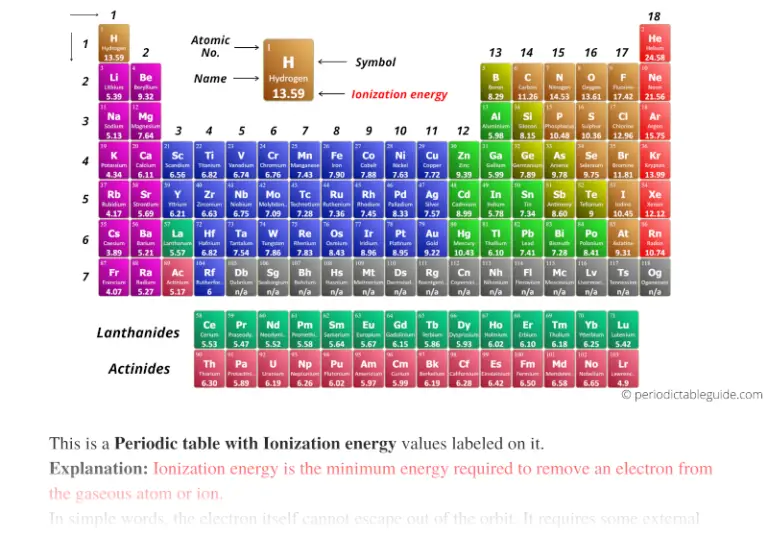
These effects tend to counter each other, and it is not obvious which will dominate. Based on periodic trends alone, it is impossible to tell which has a higher ionization energy because, as you trace the path between O and Cl, you go to the right across a row (ionization energy increases) and then down a column (ionization energy decreases). These effects sum together for an overall decrease. The first ionization energy refers to the energy it takes to remove one electron from an atom. N has a higher ionization energy than Si because as you trace the path between N and Si on the periodic table, you move down a column (ionization energy decreases) and then to the left across a row (ionization energy decreases). Ionization energy decreases as you go down a column as a result of the increasing size of orbitals with increasing n. As has a higher ionization energy than Sb because as you trace the path between As and Sb on the periodic table, you move down a column. Ionization energy increases as you go to the right due to increasing effective nuclear charge. S has a higher ionization energy than Al because as you trace the path between Al and S on the periodic table, you move to the right within the same row. These effects tend to counter each other, and it is not easy to tell which will predominate. Between Rn and Pb, the element with the higher first ionization energy is - 4. Between Mg and Ca, the element with the higher first ionization energy is - 3. 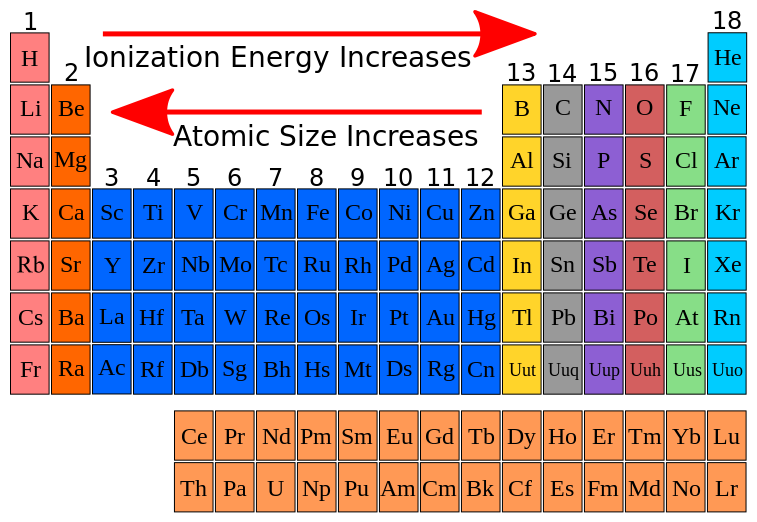
Between Kr and Br, the element with the higher first ionization energy is - 2. Based on periodic trends alone, you cannot tell which atom is larger because as you trace the path between Al and Ge you move to the right across a period (atomic size decreases) and then down a column (atomic size increases). Choose the element with the highest first ionization energy from each of the following pairs. These effects add together for an overall increase. Al atoms are larger than N atoms because as you trace the path between N and Al on the periodic table, you move down a column (atomic size increases) and then to the left across a period (atomic size increases). Because positive charge binds electrons more strongly, the second ionization energy of an element is always higher than the first. Atomic size increases as you move down a column because the outermost electrons occupy orbitals with a higher principal quantum number that are therefore larger, resulting in a larger atom. An elements second ionization energy is the energy required to remove the outermost, or least bound, electron from a 1+ ion of the element. Another deviation occurs as orbitals become more. Of Ca or Sr, the element with the higher first ionization energy is Ca. Ge atoms are larger than C atoms because as you trace the path between C and Ge on the periodic table, you move down a column. This version of the periodic table shows the first ionization energy of (IE1), in kJ/mol, of selected elements. Question: Of C or P, the element with the higher first ionization energy is C. As you move to the right across a period, the effective nuclear charge experienced by the outermost electrons increases, resulting in a smaller radius. C atoms are larger than F atoms because as you trace the path between C and F on the periodic table, you move to the right within the same period.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
